Zettalab molecular biology software unifies DNA sequence workflows—visualization and editing, plasmid construction, primer design,
sequence alignment, and translation—so teams can design and validate experiments in one place. Pair it with ELN software for compliant records across R&D.
Molecular Biology
Molecular biology software for DNA sequence editing, primer design & sequence alignment
Unify & Manage the data
Ingest, organize, and search all your molecular data
(sequences, structures, protocols, results) in one unified,
searchable platform. Eliminate spreadsheet chaos.
Increase throughput
Run molecular biology software workflows at scale: bulk cloning,
sequence alignment, and translation tools.
Collaborate Seamlessly
Securely synchronize data, protocols, and analysis within your
team. Annotate, discuss, and track project progress
effortlessly.
Visualization
Gain unparalleled visibility of your DNA or protein sequence
- Highlight promoters, tags, domains, and more with an extensive feature library
- Intuitive, interactive view that makes design and verification effortless
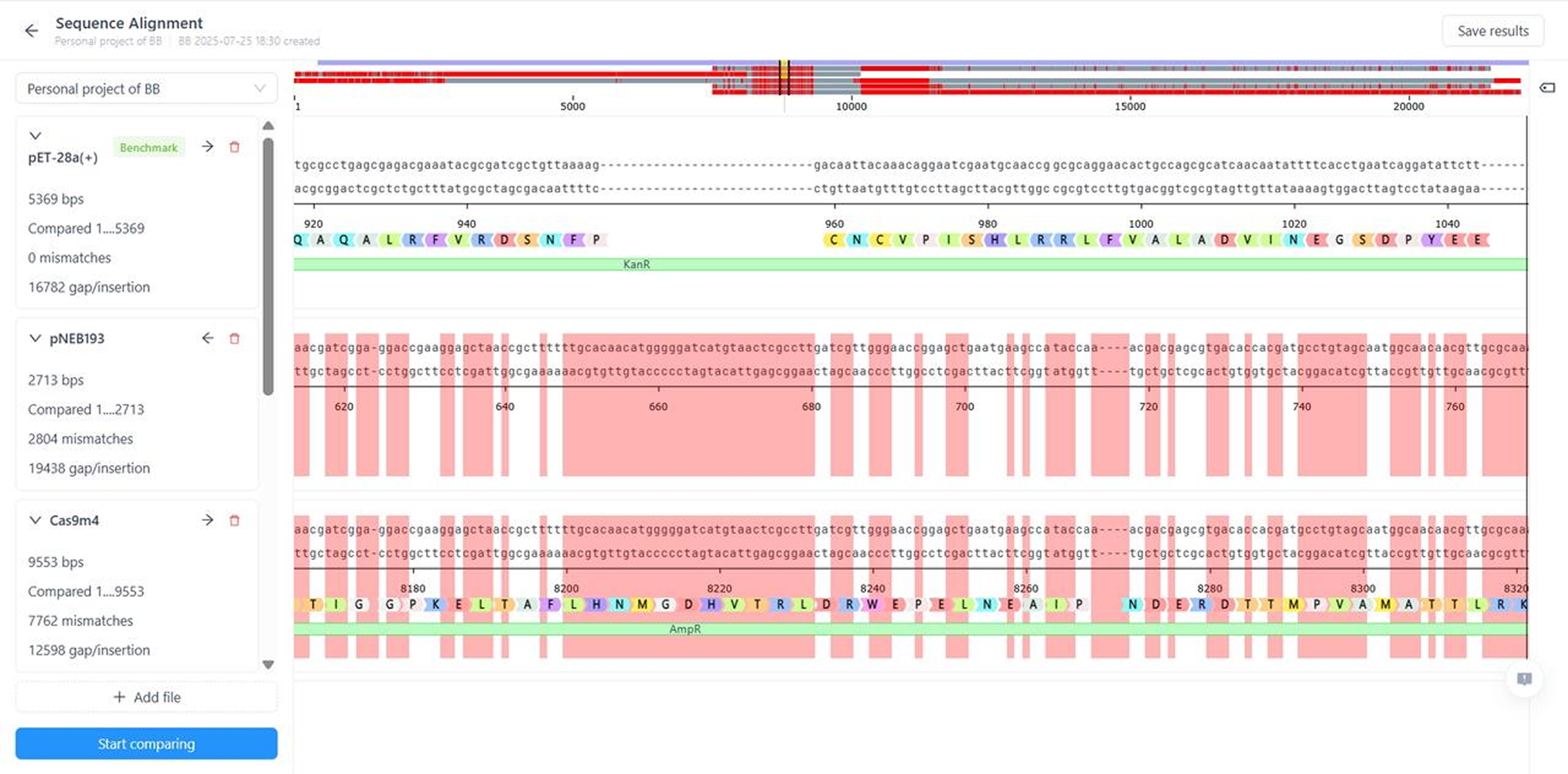
- Verify experimental results with visualized alignment of sequencing results
- Document annotations for ORFs, primers, and restriction enzyme sites
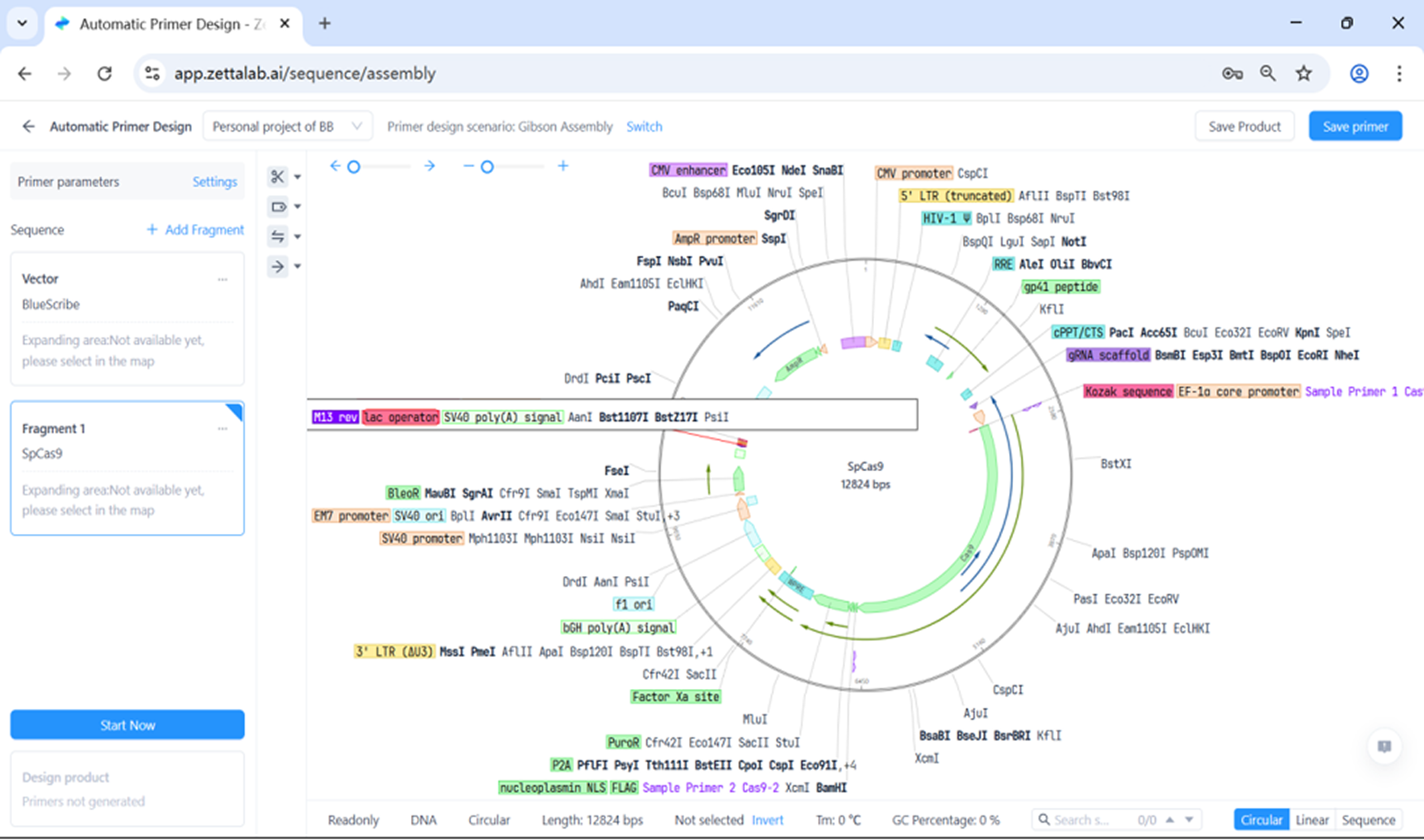
Comprehensive Molecular Design Platform
Advanced tools for molecular biology research
- Elegant, informative windows for simulating common cloning and PCR methods
- Identify and avoid common mis-steps by keeping track of details like DNA methylation and phosphorylation
- Simulate bulk cloning methods such as restriction enzyme digestion, Gibson assembly or homologous alignment
- Optimize DNA and RNA primers with a step-by-step design wizard
- Design CRISPR guide-RNAs with on-/off-target scoring
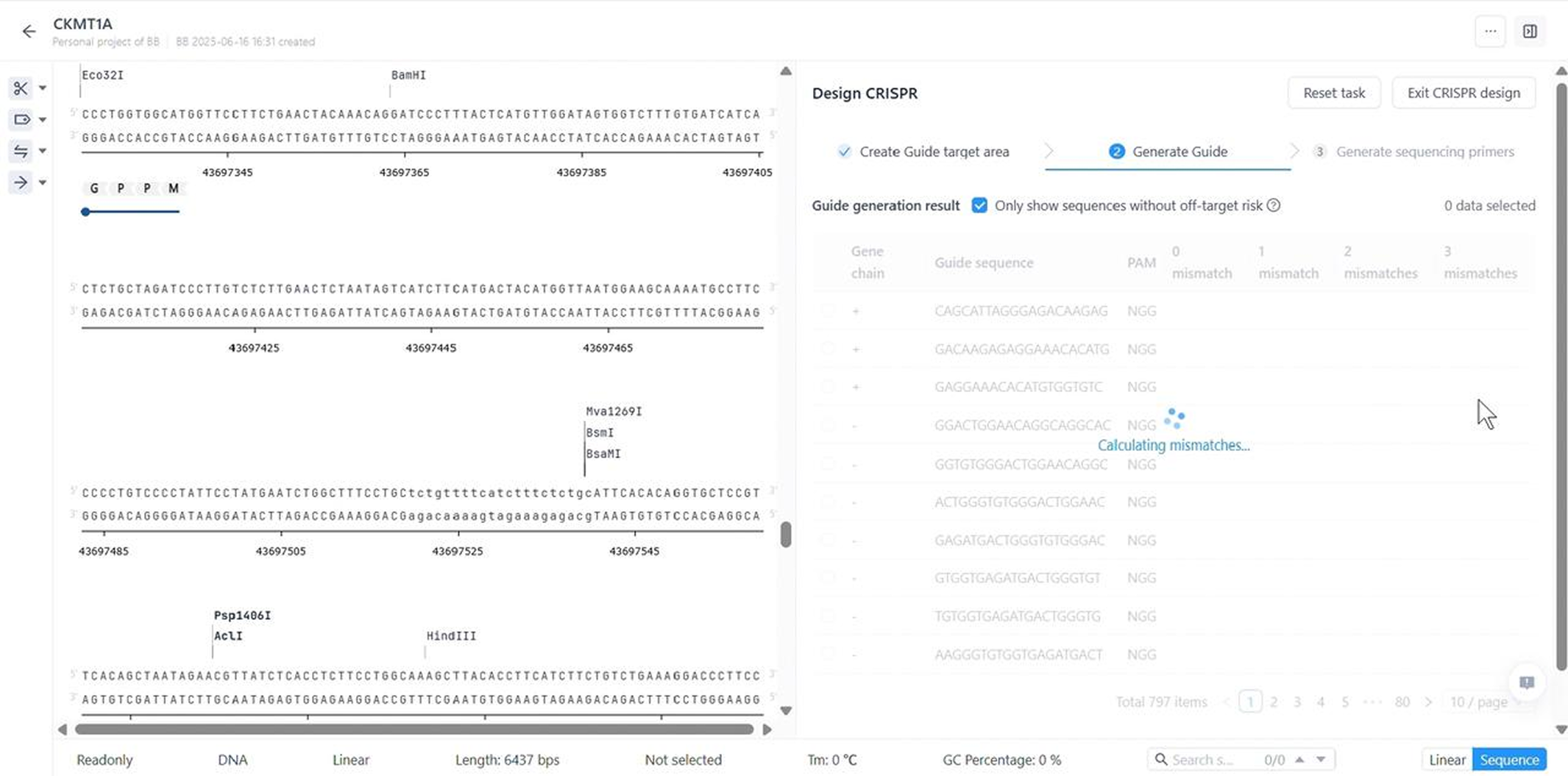
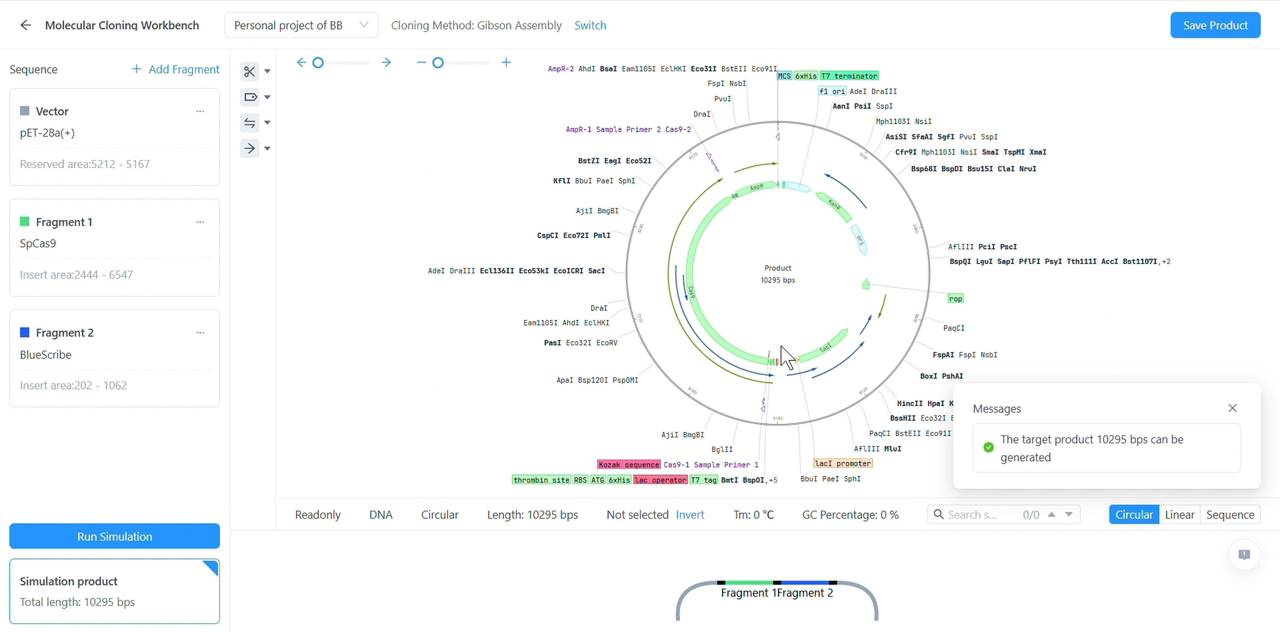
Sequence Analysis
Intelligent Sequence Analysis Tools
- Annotate DNA/RNA/protein sequences with standardized, shared libraries to ensure consistency and collaborative efficiency
- Instantly compute critical properties (e.g., molecular weight, isoelectric point, extinction coefficients) for any sequence type
- Perform single or bulk alignments using an expanded algorithm suite to optimize for speed and accuracy
- Predict and visualize 3D protein structures with industry-leading accuracy (Contact us to open the functionality)
ELN software for your electronic lab notebook
Zettalab ELN software delivers a full electronic lab notebook experience for modern labs: record, trace, and share experiment documentation seamlessly.
What is an electronic lab notebook?
An electronic lab notebook (ELN) is a digital platform
designed to replace traditional paper-based lab notebooks. It
serves as a centralized and organized space for researchers to
record, manage, and share experimental data, protocols, and
results. ELNs are equipped with features such as automated
data backup, version control, audit trails, and search
functionality, which enhance data integrity, security, and
accessibility. They also facilitate collaboration among
researchers, enabling real-time sharing of information and
reducing the risk of data loss.
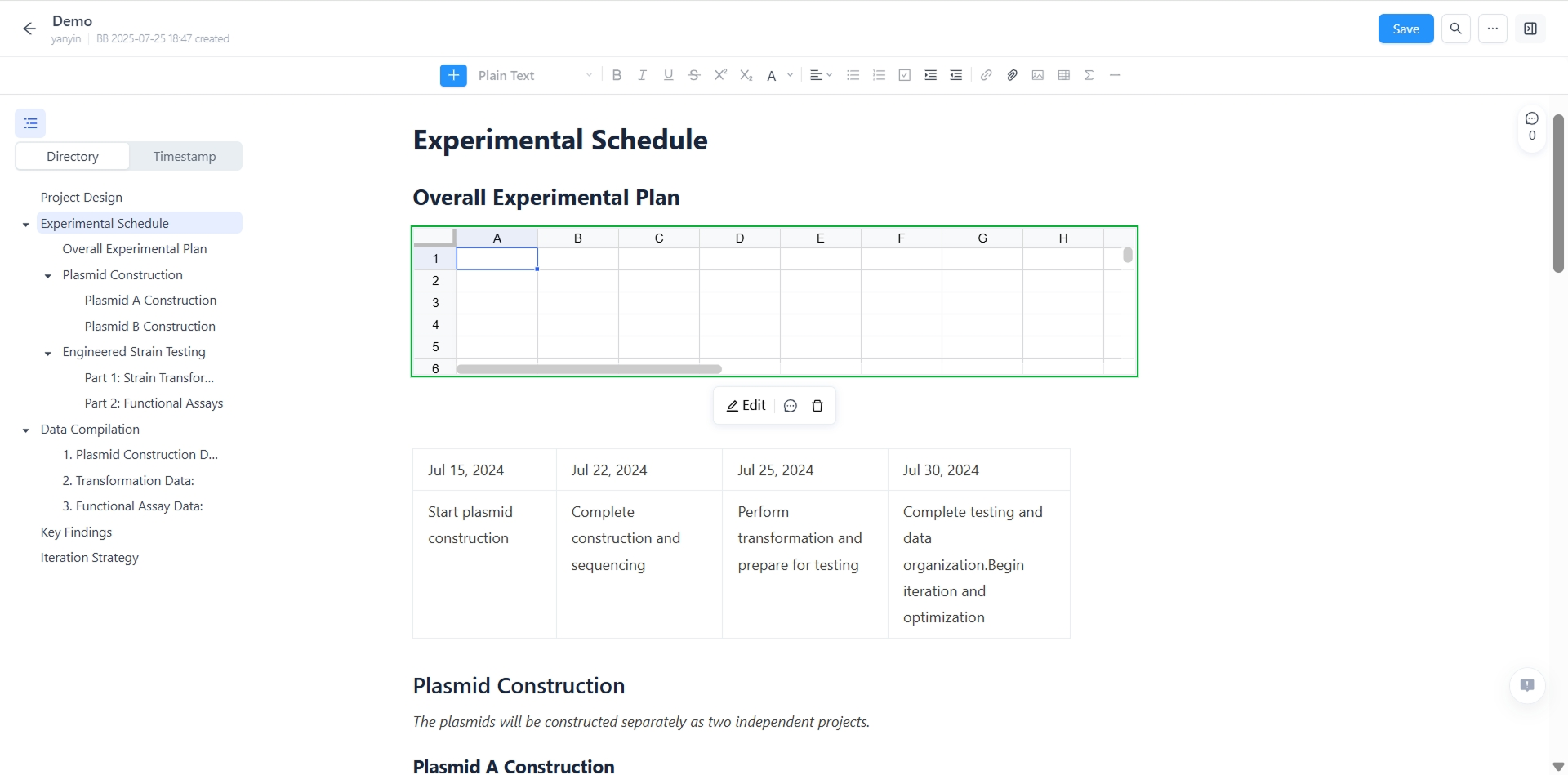
Data and Template Documentation
Experiment documentation software for structured lab data
- Create templates and procedures to streamline experiment documentation software workflows—standardize data management, retrieval, and sharing of results. Tailored checklists to define tasks, permissions, and resources.
- Search inside lab work entries for easy data retrieval.
- Tailored checklists to define tasks, permissions, and resources.
Team Collaboration
Enhanced collaboration for research teams
- Improve cross-team communication and collaboration
- Improved real-time knowledge sharing across teams to strengthen collaboration and eliminate silos.
- Remind or notify team members.
- Associate resources with tasks and projects.
- Set read and write access levels for project management.
Safeguard Data Reliability
Ensure data integrity and reliability
- Implement standardized frameworks to ensure unified recording, collaborative sharing, and strategic decision support.
- Enhance accuracy by automating manual steps.
- Standardized knowledge archiving and access.

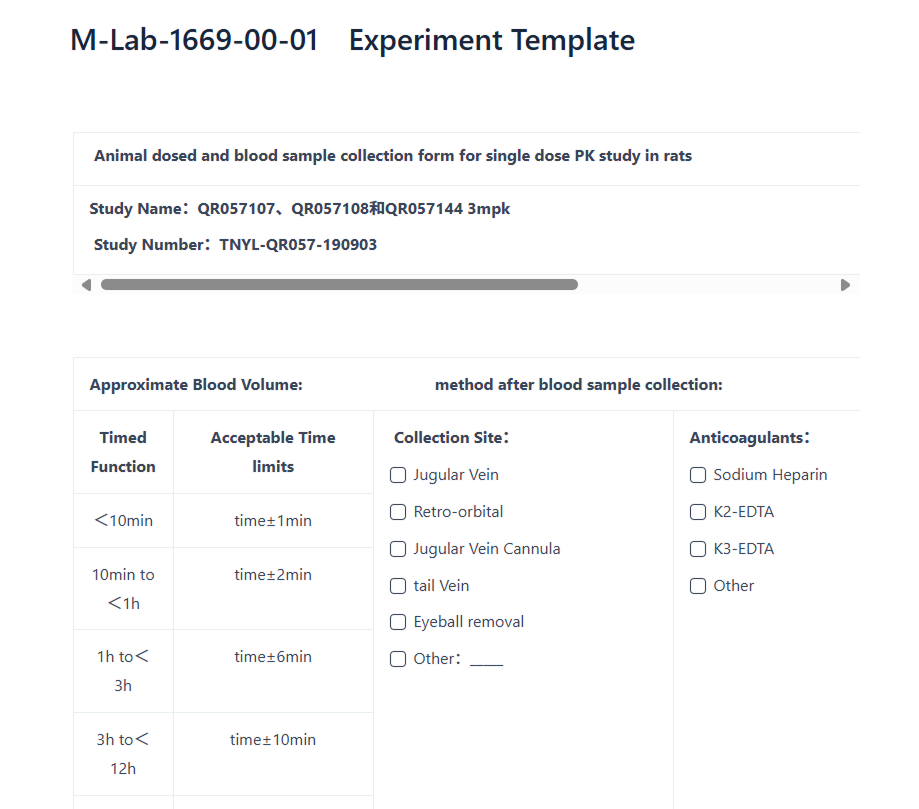
Protected Data Storage
Secure and reliable data storage solutions
- Securely centralize, document, and archive all information within a cloud-based repository.
- Access your data/information from any location.
- Reliable methodologies with a verifiable history of actions.
- Prevent data loss in on-premises systems.
Achieve Compliance Requirements
Meet regulatory and compliance standards
- Build compliance-driven SOPs for organizational governance.
- Audit revision tracking and access controls.
- Comply with 21 CFR Part 11 regulations.
- Support regulatory compliance through complete audit trails and demonstrable data history.
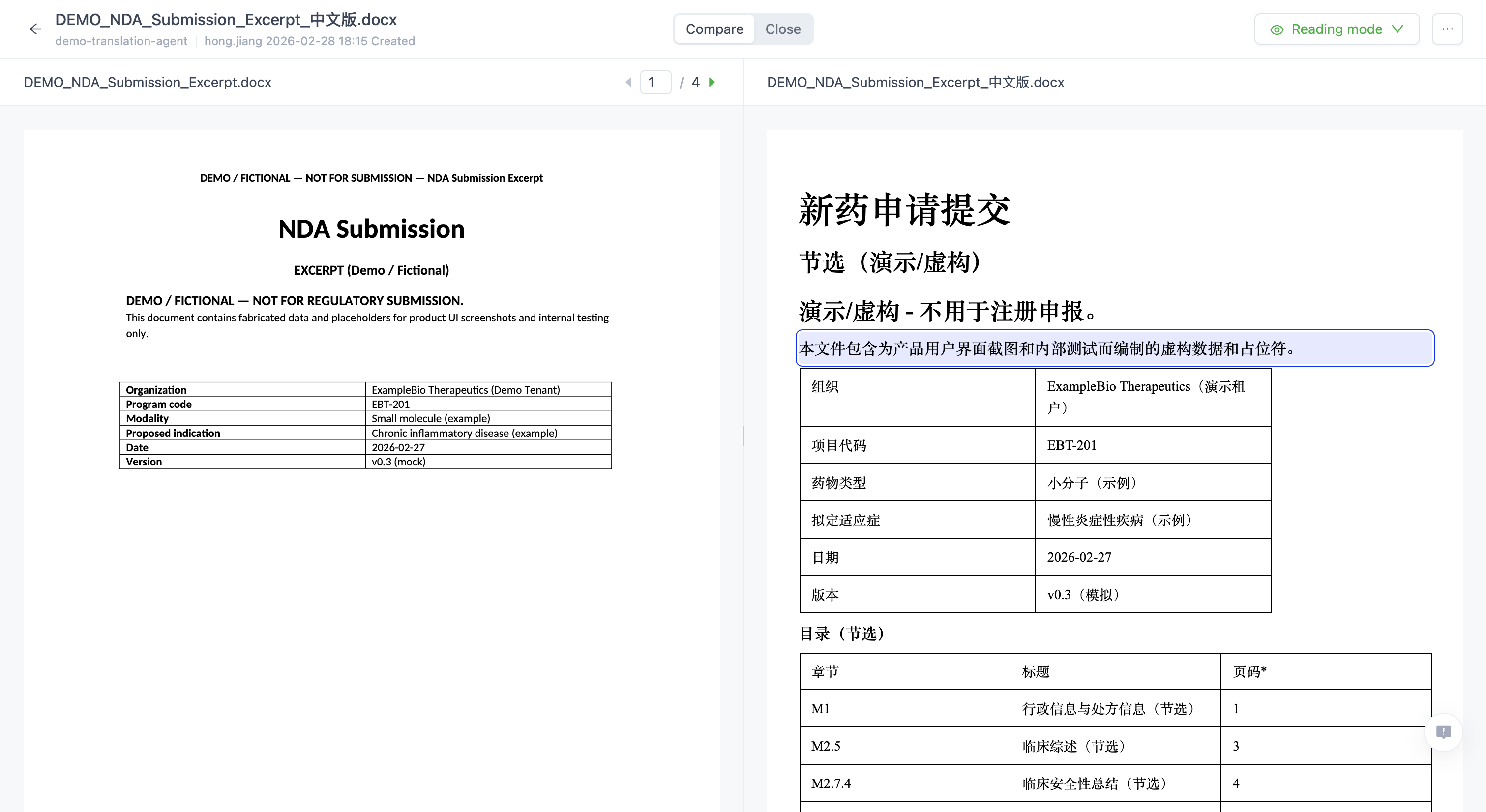
AI Translation Agent
Domain-Specific AI Translation for Global Biopharma Registration
Zettalab AI Translation Agent is an enterprise-grade AI system built for regulatory document translation in life sciences. Powered by vertical LLM foundation models, it delivers high-accuracy translation, terminology consistency, format preservation, and full compliance traceability for global biopharma registration. Designed for pharmaceutical companies, CROs, and research institutions to translate complex regulatory documents securely and at scale.
High-Accuracy Domain Translation
Fine-tuned life science language models and a regulatory-aware translation engine deliver high-precision output, while cross-model validation reduces hallucination and context-length optimization supports long dossiers, resulting in translation accuracy above 97%.
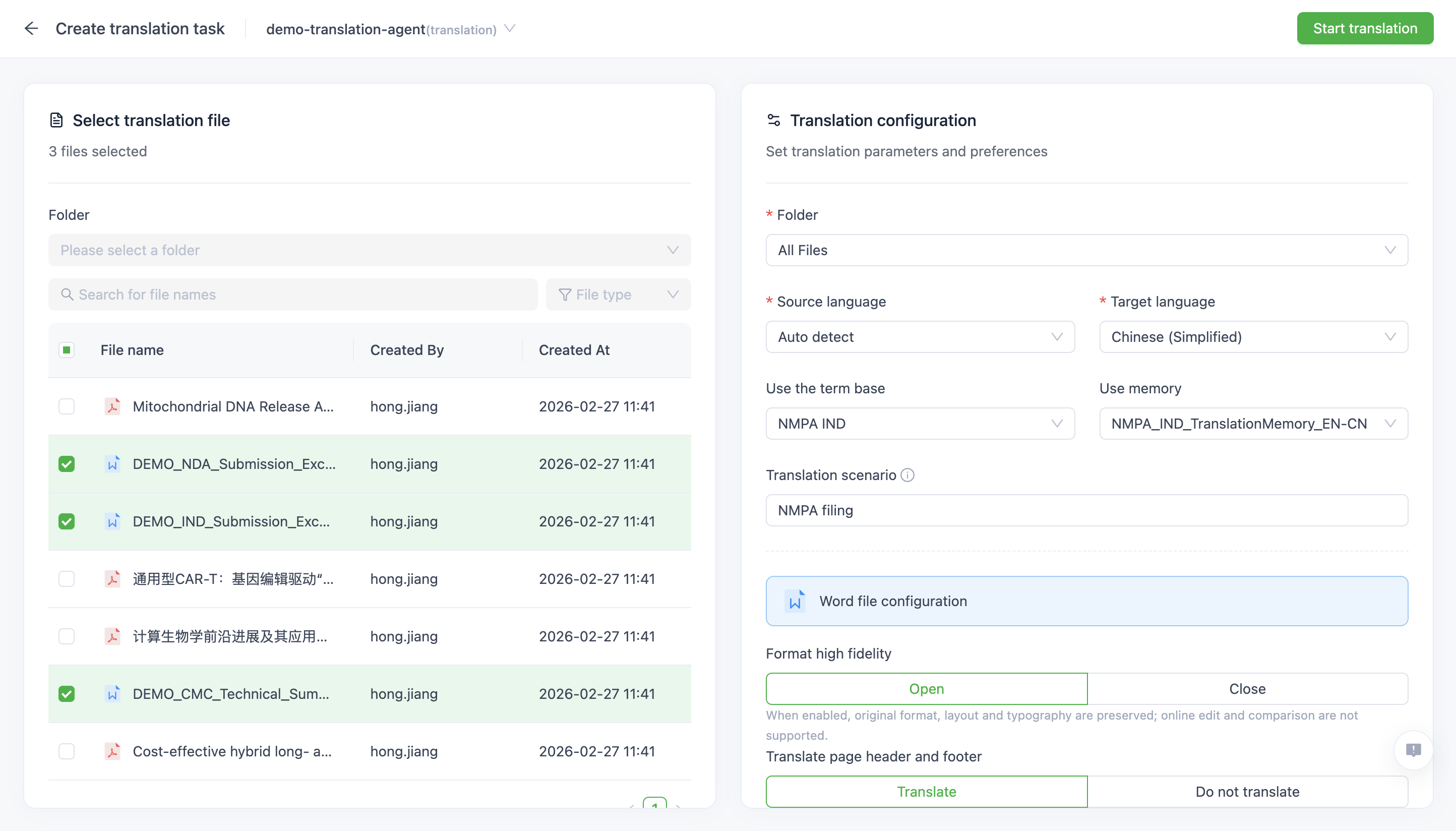
Intelligent Document Processing
Automatic document segmentation and context-aware translation support both batch and real-time workflows across Word, PDF, scanned files, and tables, and are optimized for long-form regulatory dossiers including IND, NDA, CTA, and BLA submissions.
Format Preservation & Structural Alignment
Structure-aware translation preserves over 90% of original document formatting, aligns source and target paragraphs, and exports in original file formats to minimize manual reformatting and reduce downstream review effort.
Terminology & Translation Memory
A built-in enterprise termbase with terminology extraction, validation, domain glossary integration, and private translation memory ensures consistent language across regulatory submissions, clinical documents, and cross-team collaboration.
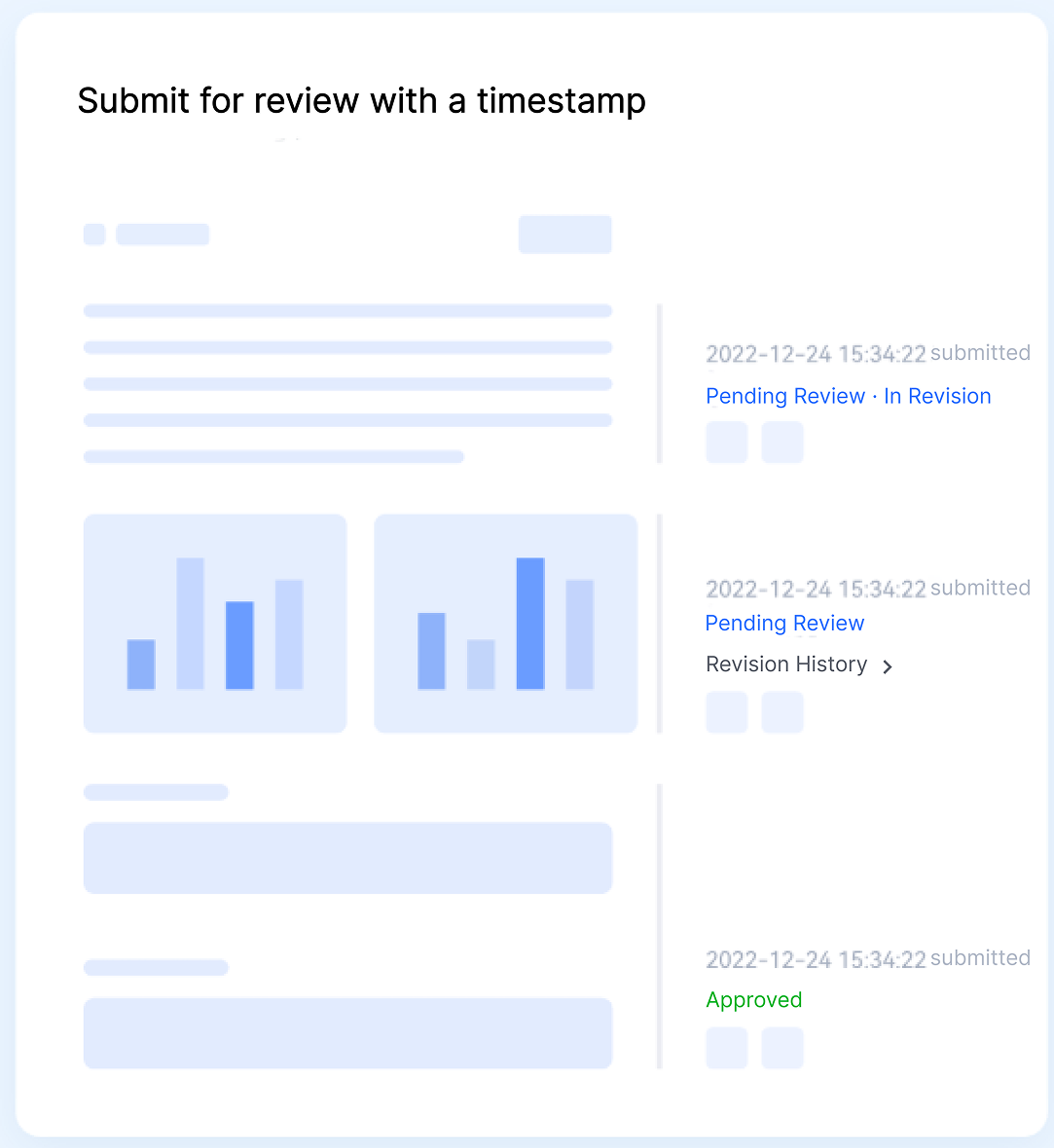
Traceability & Audit Control
Built for compliance-driven environments, the platform supports source-target comparison, technical content tagging, selective retranslation, and full audit logging to provide complete traceability and stronger quality control throughout translation cycles.
Seamless User Experience
From Ingestion to Compliance-Ready Export
- Structured document ingestion for regulatory files
- Domain-specific terminology control
- AI translation with validation review
- Source-target alignment and format preservation
- Compliance-ready export with full traceability
Enterprise AI Architecture
Enterprise Level AI Translation Infrastructure
- Domain-specific foundation models enhanced with fine-tuned LLMs, prompt orchestration, and vectorized knowledge retrieval.
- End-to-end translation workflow integrating QA validation, terminology control, and project management automation.
- Enterprise security architecture with identity control, RBAC, and full audit traceability.
- Encrypted data management with structured metadata governance and secure storage.
- Deployable on-premise or in private cloud environments to meet pharmaceutical compliance standards.
Diverse User Scenarios
Speed the Pipeline Registration
- Accelerates regulatory submission translation for IND, NDA, and BLA documentation across global markets.
- Ensures precision and consistency in clinical trial and investigator documentation.
- Supports seamless multilingual collaboration in international research environments.
- Strengthens CRO and enterprise documentation with standardized, compliant translation workflows.
- Enhances regulatory communication efficiency throughout the product lifecycle.
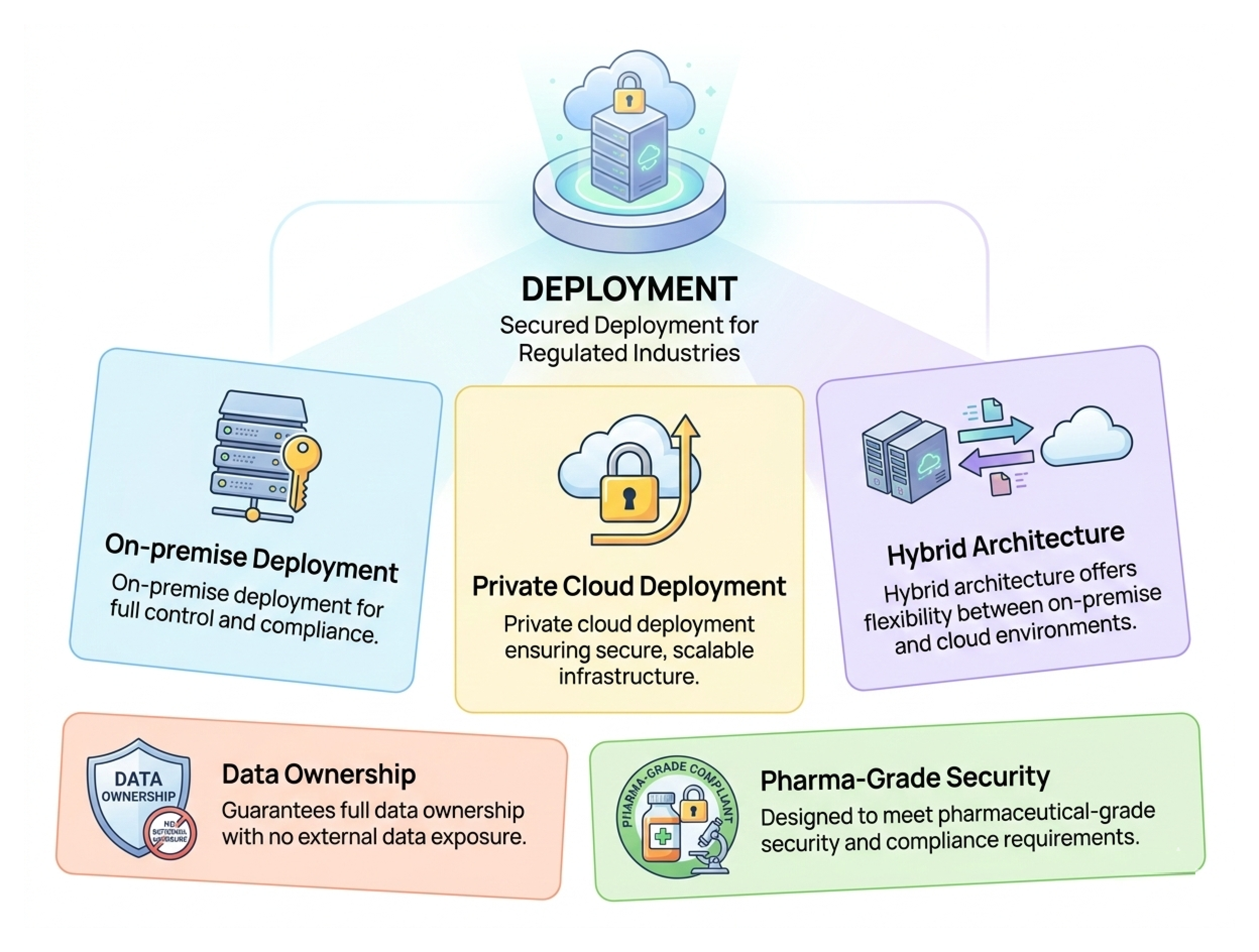
Secured Deployment
Secure Deployment for Regulated Industries
- On-premise deployment for full control and compliance.
- Private cloud deployment ensuring secure, scalable infrastructure.
- Hybrid architecture offers flexibility between on-premise and cloud environments.
- Guarantees full data ownership with no external data exposure.
- Designed to meet pharmaceutical-grade security and compliance requirements.
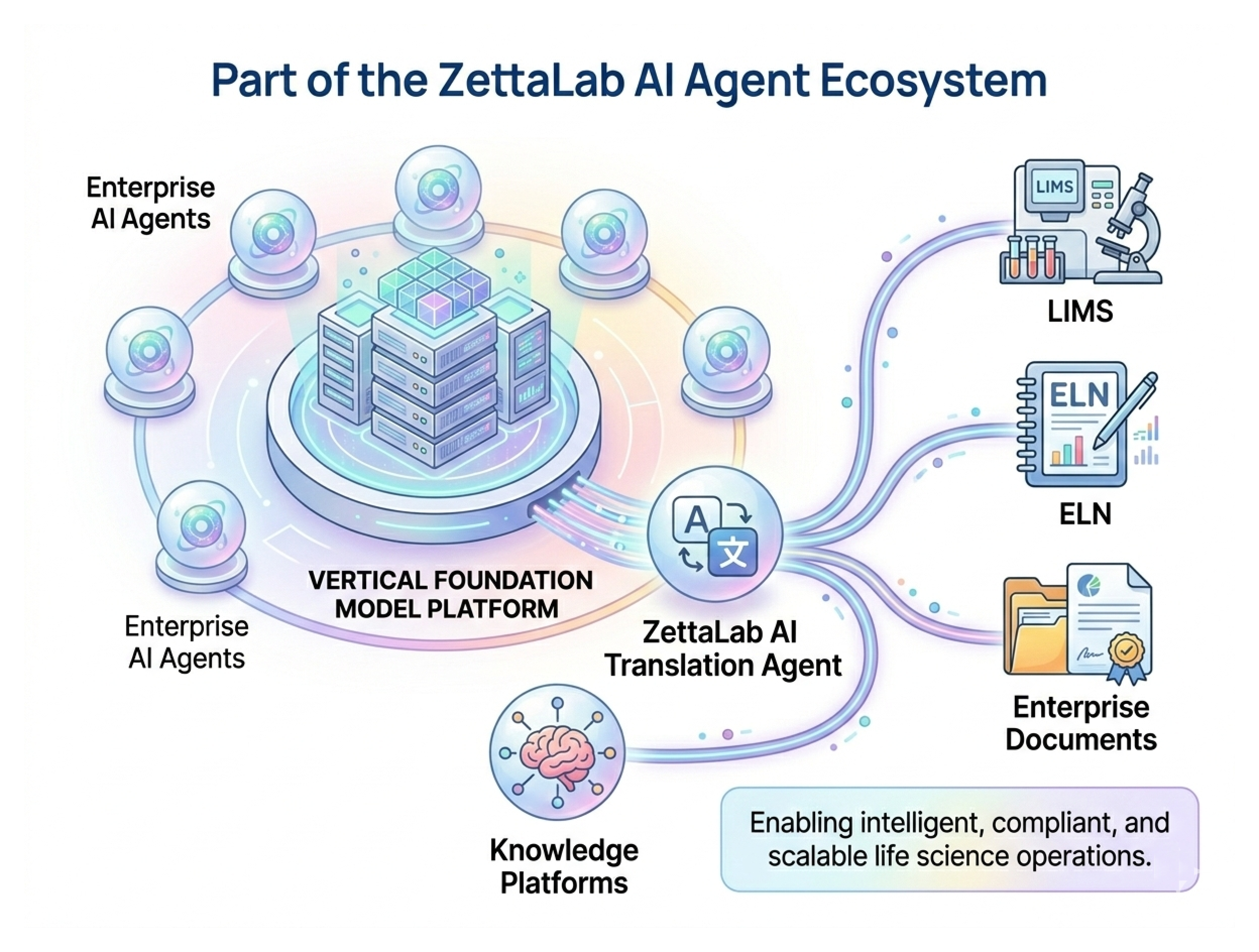
Part of the Zettalab AI Agent Ecosystem
One Vertical Foundation Model Platform + N Enterprise AI Agents
Zettalab AI Translation Agent integrates seamlessly with:
- LIMS
- ELN
- Enterprise document systems
- Knowledge management platforms
Enabling intelligent, compliant, and scalable life science operations.