How Do Tamper-Proof Experiment Records Transform Laboratory Data Integrity and Compliance?
How Do Tamper-Proof Experiment Records Transform Laboratory Data Integrity and Compliance?
Meta Description: Learn how tamper-proof experiment records ensure data integrity, meet GLP/GMP compliance standards, and protect research organisations from data manipulation risks.
Why Laboratory Data Integrity Matters More Than Ever
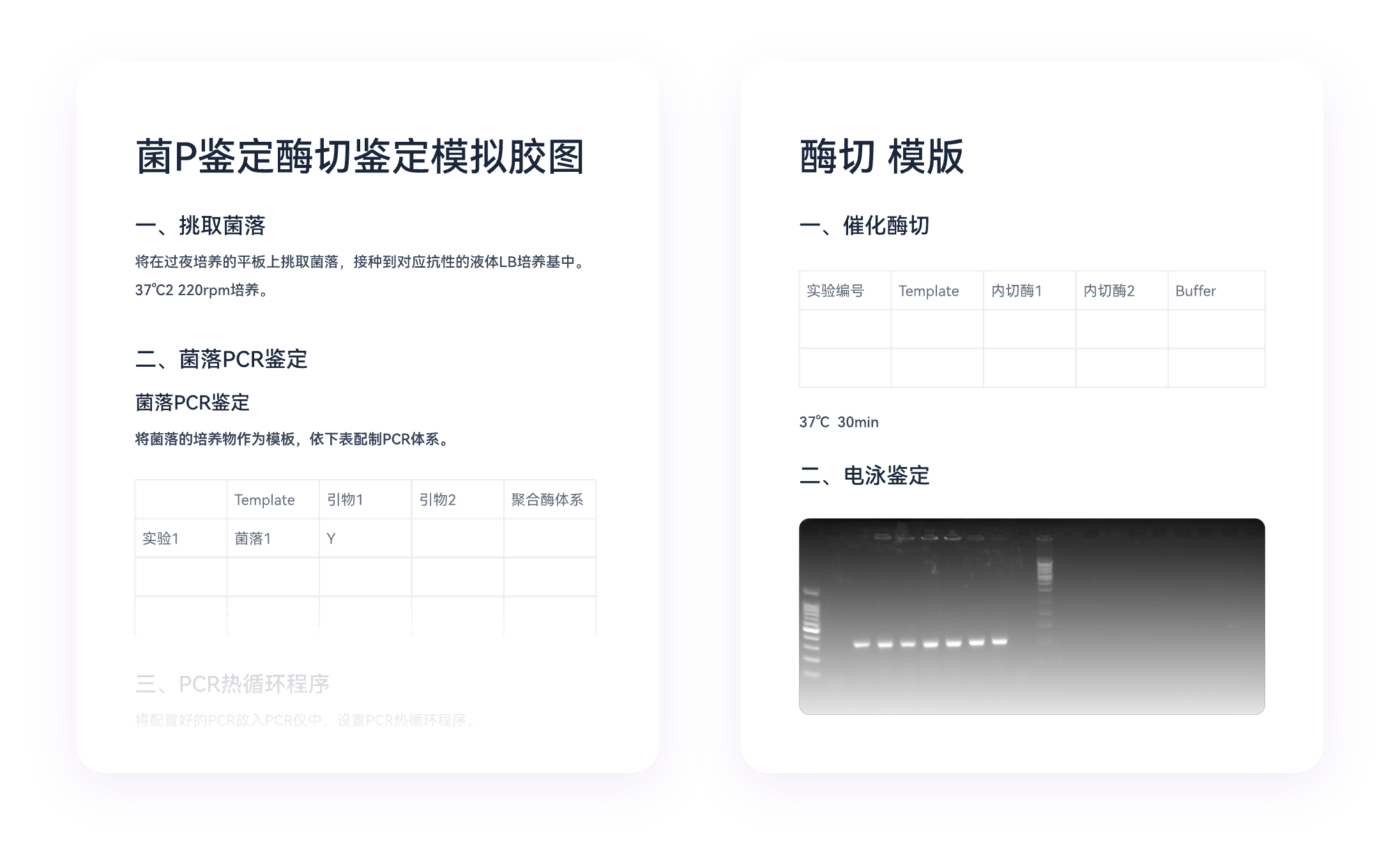
Modern research environments generate enormous volumes of experimental data that must remain trustworthy throughout the research lifecycle. Tamper-proof experiment records have emerged as essential tools for maintaining the credibility and reproducibility of scientific findings.
Laboratory managers and principal investigators increasingly recognise that conventional paper notebooks and basic digital files cannot guarantee the integrity researchers require. Without robust safeguards, experimental data becomes vulnerable to accidental modifications, deliberate manipulation, or loss.
The Growing Importance of Data Security in Research
Critical factors driving the need for secure records:
- Regulatory requirements mandating documented evidence trails
- Intellectual property protection for proprietary research
- Reproducibility standards in peer-reviewed publications
- Audit requirements from funding agencies and sponsors
- Legal defensibility of research outcomes and patents
Research organisations implementing tamper-proof experiment records report significant improvements in compliance outcomes and reduced audit findings.
What Makes Experiment Records Tamper-Proof
Core Security Features
Effective tamper-proof experiment records incorporate multiple layers of protection that work together to ensure data authenticity and permanence.
Essential security elements:
- Cryptographic hashing: Mathematical algorithms that detect any changes to original files
- Digital signatures: Verifiable proof of document authorship and timing
- Audit trails: Complete logs recording every access and modification attempt
- Immutable storage: Systems preventing retroactive alterations to stored data
- Access controls: Role-based permissions limiting who can view or modify records
These features combine to create experiment records that researchers can trust completely for critical regulatory submissions and scientific publications.
Comparison of Record Protection Methods
| Method | Security Level | Cost | Ease of Implementation | Regulatory Acceptance |
|---|---|---|---|---|
| Paper notebooks with signatures | Low | Low | High | Limited |
| Basic electronic files | Very Low | Very Low | Very High | None |
| Password-protected documents | Low | Low | High | Limited |
| Dedicated ELN with audit trails | Medium-High | Medium | Medium | Good |
| Blockchain-backed records | Very High | High | Low-Medium | Excellent |
| Compliant electronic systems | High | Medium-High | Medium | Excellent |
How Tamper-Proof Experiment Records Support GLP and GMP Compliance
Meeting Regulatory Expectations
Good Laboratory Practice (GLP) and Good Manufacturing Practice (GMP) regulations require organisations to maintain accurate and complete records of all experimental activities. Tamper-proof experiment records provide the documentary evidence regulators demand.
GLP/GMP record requirements addressed:
- Complete documentation of experimental procedures and observations
- Chronological recording with date and time stamps
- Identification of individuals performing and witnessing activities
- Protection against unauthorised modifications
- Long-term preservation and accessibility
Laboratory quality managers implementing tamper-proof experiment records consistently report smoother regulatory inspections and fewer compliance deficiencies.
FDA and EMA Guidance Alignment
Regulatory agencies worldwide have issued guidance emphasising the importance of electronic record integrity. The FDA's 21 CFR Part 11 and EU Annex 11 specifically address requirements for electronic signatures and tamper-evident record-keeping.
Key regulatory expectations:
| Requirement | How Tamper-Proof Records Help |
|---|---|
| Audit trail verification | Automated logging of all record interactions |
| Electronic signature validity | Cryptographic verification capabilities |
| Long-term accessibility | Standardised formats ensuring future readability |
| System validation documentation | Built-in compliance reporting features |
| Change control evidence | Complete modification history with justification |
What Types of Laboratories Benefit Most from Secure Records
Pharmaceutical Research and Development
Drug development organisations conducting preclinical and clinical studies rely heavily on tamper-proof experiment records to support regulatory submissions. The stakes in pharmaceutical research demand nothing less than absolute data integrity.
Applications in pharmaceutical laboratories:
- Bioanalytical method development and validation
- Stability testing programmes
- Manufacturing process development
- Quality control testing documentation
- Clinical trial sample analysis records
Academic Research Institutions
University laboratories increasingly adopt tamper-proof experiment records to meet journal requirements for data availability and research integrity standards.
Academic use cases:
- Graduate student thesis research documentation
- Multi-institutional collaborative projects
- Longitudinal studies spanning multiple years
- Publicly funded research grant requirements
- Open science data sharing initiatives
Contract Research Organisations
CROs serving multiple clients must demonstrate robust data handling practices to maintain trust and competitive advantage. Tamper-proof experiment records provide third-party verification capabilities clients increasingly demand.
How to Implement Tamper-Proof Experiment Records in Your Laboratory
Assessment and Planning Phase
Successful implementation begins with thorough evaluation of current record-keeping practices and identification of specific integrity requirements.
Initial assessment checklist:
- Document current record storage methods and their vulnerabilities
- Identify regulatory requirements applicable to your laboratory
- Evaluate existing electronic laboratory notebook capabilities
- Survey staff on current record-keeping challenges
- Define minimum security requirements for different record types
- Establish budget and timeline parameters for implementation
Technology Selection Criteria
Choosing appropriate technology solutions requires careful consideration of laboratory-specific needs and future scalability.
Key evaluation factors:
| Criterion | Questions to Consider |
|---|---|
| Regulatory compliance | Does the system meet applicable GLP/GMP requirements? |
| Integration capability | How well does it connect with existing laboratory instruments? |
| User experience | Will researchers adopt the system without excessive training? |
| Vendor stability | Is the provider likely to support the system long-term? |
| Cost structure | Are licensing and maintenance costs sustainable? |
| Security certification | Has the system undergone independent security assessment? |
Implementation Steps for Success
Phased rollout approach:
- Pilot phase: Test system with selected projects or research groups
- Training phase: Develop comprehensive user training programmes
- Validation phase: Document system validation for regulated activities
- Broad deployment: Extend system access across the organisation
- Continuous improvement: Regular review and optimisation of workflows
What Are the Common Challenges in Maintaining Data Integrity
Human Factors and Resistance to Change
Even the most sophisticated tamper-proof experiment records cannot succeed without researcher buy-in and proper training. Understanding common adoption barriers helps organisations address them proactively.
Frequently encountered challenges:
- Perceived increase in administrative burden
- Concerns about system complexity
- Reluctance to abandon familiar paper-based methods
- Insufficient training on new electronic systems
- Lack of clear organisational commitment to implementation
Technical Implementation Obstacles
Laboratory environments present unique technical challenges that can compromise tamper-proof experiment records if not properly addressed.
Technical challenges and solutions:
| Challenge | Potential Impact | Mitigation Strategy |
|---|---|---|
| Legacy instrument integration | Data gaps in electronic records | Middleware solutions or manual entry protocols |
| Network connectivity issues | Delayed record synchronisation | Offline-capable systems with sync functionality |
| Storage capacity limitations | Premature data archiving | Cloud-based storage with scalability |
| System performance degradation | User frustration and workarounds | Regular system monitoring and optimisation |
| Cybersecurity threats | Data breach or corruption | Comprehensive security framework implementation |
Why Blockchain Technology Enhances Record Integrity
Distributed Ledger Applications
Blockchain technology provides unprecedented capabilities for creating truly tamper-proof experiment records through decentralised verification mechanisms.
Blockchain advantages for laboratory records:
- Immutable transaction history preventing retroactive modifications
- Distributed consensus eliminating single points of failure
- Transparent verification enabling third-party validation
- Timestamped entries providing indisputable chronological evidence
- Reduced reliance on trusted intermediaries
Practical Blockchain Implementations
Several emerging platforms specifically target scientific research applications, offering purpose-built solutions for laboratory environments.
Current blockchain applications in research:
- Scientific data publication and verification
- Intellectual property timestamping
- Supply chain tracking for laboratory materials
- Multi-site collaborative research coordination
- Research integrity certification
How Tamper-Proof Records Protect Intellectual Property
Establishing Priority and Ownership
Research organisations investing in innovative discoveries need reliable methods for demonstrating when discoveries occurred and who contributed to them. Tamper-proof experiment records provide legally defensible evidence.
IP protection mechanisms:
- Verifiable creation timestamps establishing priority dates
- Clear attribution of inventor contributions
- Complete documentation supporting patent applications
- Evidence chain for potential litigation scenarios
- Due diligence documentation for licensing negotiations
Supporting Technology Transfer Activities
When research organisations license technologies or spin out companies, comprehensive documentation becomes essential for valuation and due diligence processes.
Technology transfer benefits:
| Stage | How Secure Records Help |
|---|---|
| Initial disclosure | Complete invention documentation |
| Patent filing | Priority date establishment |
| Valuation | Comprehensive development history |
| Due diligence | Verifiable data integrity evidence |
| Licensing | Clear ownership and attribution records |
What Best Practices Ensure Long-Term Record Integrity
Organisational Culture and Training
Technical systems alone cannot guarantee data integrity without supporting organisational practices and researcher commitment.
Cultural best practices:
- Leadership commitment to data integrity as a core value
- Regular training on record-keeping requirements and techniques
- Clear accountability for data quality at all organisational levels
- Open discussion of integrity challenges without blame culture
- Recognition and reward for exemplary record-keeping practices
Technical Maintenance and Monitoring
Tamper-proof experiment records require ongoing technical attention to maintain their protective capabilities over time.
Technical maintenance requirements:
- Regular security updates and patches
- Periodic system performance reviews
- Backup verification and disaster recovery testing
- Access control audits and permission reviews
- Long-term format migration planning
Monitoring activities:
| Activity | Frequency | Purpose |
|---|---|---|
| Security scan | Continuous | Detect unauthorised access attempts |
| Audit trail review | Weekly | Identify unusual patterns or anomalies |
| System health check | Daily | Ensure availability and performance |
| Backup verification | Monthly | Confirm data recovery capabilities |
| Access control review | Quarterly | Maintain appropriate permission levels |
How Cost-Effective Are Tamper-Proof Experiment Records
Return on Investment Considerations
While implementing tamper-proof experiment records requires upfront investment, organisations typically realise substantial returns through improved efficiency and reduced risk exposure.
Cost-benefit analysis factors:
- Reduced regulatory audit findings and associated remediation costs
- Faster preparation of regulatory submissions
- Lower risk of research integrity investigations
- Improved collaboration efficiency across research teams
- Enhanced organisation reputation and competitive positioning
Comparative Cost Structures
Different approaches to tamper-proof experiment records carry varying cost profiles that organisations must evaluate against their specific needs.
Approximate annual costs per user:
| Solution Type | Small Lab (<10 users) | Medium Lab (10-50 users) | Large Lab (>50 users) |
|---|---|---|---|
| Basic electronic notebook | $200-500 | $150-400 | $100-300 |
| Compliant ELN system | $500-1,500 | $400-1,200 | $300-1,000 |
| Enterprise platform | $1,000-3,000 | $800-2,500 | $500-2,000 |
| Blockchain-backed solution | $1,500-5,000 | $1,200-4,000 | $800-3,000 |
Conclusion: Building Trust Through Verified Data Integrity
Tamper-proof experiment records represent essential infrastructure for modern research organisations committed to scientific integrity and regulatory compliance. By implementing robust technical safeguards supported by appropriate organisational practices, laboratories can ensure their experimental data remains trustworthy throughout the research lifecycle.
Whether driven by regulatory requirements, publication standards, or internal quality objectives, investment in tamper-proof experiment records delivers substantial returns through reduced compliance risk, improved efficiency, and enhanced organisational reputation. As regulatory expectations continue evolving and scientific reproducibility concerns persist, organisations prioritising data integrity position themselves for long-term success.
The path forward requires thoughtful technology selection, comprehensive training programmes, and sustained organisational commitment. However, the resulting benefits—in regulatory confidence, research credibility, and operational efficiency—make tamper-proof experiment records an investment well worth making for any serious research organisation.